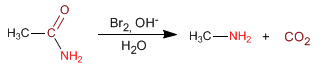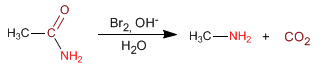-
Details
-
Germán Fernández
-
AMINES THEORY
-
Hits: 41579
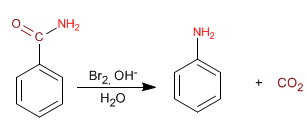
Amides are converted to amines, with one less carbon, by treatment with bromine in a basic medium. This reaction is known as the Hofmann rearrangement.
The amide reacts with bromine in a basic medium forming an N-bromoamide, which regroups to isocyanate. Hydrolysis of the isocyanate produces carbamic acid, which decarboxylates to give the amine.